Abstract
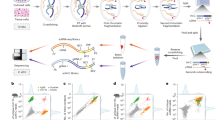
Although single-gene loss-of-function analyses can identify components of particular processes, important molecules are missed owing to the robustness of biological systems. Here we show that large-scale RNAi screening for suppression interactions with functionally related mutants greatly expands the repertoire of genes known to act in a shared process and reveals a new layer of functional relationships. We performed RNAi screens for 17 Caenorhabditis elegans cell polarity mutants, generating the most comprehensive polarity network in a metazoan, connecting 184 genes. Of these, 72% were not previously linked to cell polarity and 80% have human homologues. We experimentally confirmed functional roles predicted by the network and characterized through biophysical analyses eight myosin regulators. In addition, we discovered functional redundancy between two unknown polarity genes. Similar systematic genetic interaction screens for other biological processes will help uncover the inventory of relevant genes and their patterns of interactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lehner, B., Crombie, C., Tischler, J., Fortunato, A. & Fraser, A. G. Systematic mapping of genetic interactions in Caenorhabditis elegans identifies common modifiers of diverse signalling pathways. Nat. Gen. 38, 896–903 (2006).
Byrne, A. B. et al. A global analysis of genetic interactions in Caenorhabditis elegans. J. Biol. 6, 8 (2007).
Sandmann, T. & Boutros, M. Screens, maps networks: from genome sequences to personalized medicine. Curr. Opin. Genet. Dev. 22, 36–44 (2012).
Costanzo, M. et al. The genetic landscape of a cell. Science 327, 425–431 (2010).
Labbe, J. C., Pacquelet, A., Marty, T. & Gotta, M. A genomewide screen for suppressors of par-2 uncovers potential regulators of PAR protein-dependent cell polarity in Caenorhabditis elegans. Genetics 174, 285–295 (2006).
O’Rourke, S. M., Dorfman, M. D., Carter, J. C. & Bowerman, B. Dynein modifiers in C. elegans: light chains suppress conditional heavy chain mutants. PLoS Genet. 3, 1339–1354 (2007).
Dorfman, M., Gomes, J. E., O’Rourke, S. & Bowerman, B. Using RNA interference to identify specific modifiers of a temperature-sensitive, embryonic-lethal mutation in the Caenorhabditis elegans ubiquitin-like Nedd8 protein modification pathway E1-activating gene rfl-1. Genetics 182, 1035–1049 (2009).
Coradini, D., Casarsa, C. & Oriana, S. Epithelial cell polarity and tumorigenesis: new perspectives for cancer detection and treatment. Acta Pharmacol. Sin. 32, 552–564 (2011).
St Johnston, D. & Ahringer, J. Cell polarity in eggs and epithelia: parallels and diversity. Cell 141, 757–774 (2010).
Gonczy, P. Mechanisms of asymmetric cell division: flies and worms pave the way. Nat. Rev. Mol. Cell Biol. 9, 355–366 (2008).
Motegi, F. et al. Microtubules induce self-organization of polarized PAR domains in Caenorhabditis elegans zygotes. Nat. Cell Biol. 13, 1361–1367 (2011).
Goldstein, B. & Hird, S. N. Specification of the anteroposterior axis in Caenorhabditis elegans. Development 122, 1467–1474 (1996).
Cowan, C. R. & Hyman, A. A. Centrosomes direct cell polarity independently of microtubule assembly in C. elegans embryos. Nature 431, 92–96 (2004).
Tsai, M. C. & Ahringer, J. Microtubules are involved in anterior–posterior axis formation in C. elegans embryos. J. Cell Biol. 179, 397–402 (2007).
Zonies, S., Motegi, F., Hao, Y. & Seydoux, G. Symmetry breaking and polarization of the C. elegans zygote by the polarity protein PAR-2. Development 137, 1669–1677 (2010).
Munro, E., Nance, J. & Priess, J. R. Cortical flows powered by asymmetrical contraction transport PAR proteins to establish and maintain anterior–posterior polarity in the early C. elegans embryo. Dev. Cell 7, 413–424 (2004).
Motegi, F. & Sugimoto, A. Sequential functioning of the ECT-2 RhoGEF, RHO-1 and CDC-42 establishes cell polarity in Caenorhabditis elegans embryos. Nat. Cell Biol. 8, 978–985 (2006).
Schonegg, S. & Hyman, A. A. CDC-42 and RHO-1 coordinate acto-myosin contractility and PAR protein localization during polarity establishment in C. elegans embryos. Development 133, 3507–3516 (2006).
Boyd, L., Guo, S., Levitan, D., Stinchcomb, D. T. & Kemphues, K. J. PAR-2 is asymmetrically distributed and promotes association of P granules and PAR-1 with the cortex in C. elegans embryos. Development 122, 3075–3084 (1996).
Watts, J. L. et al. par-6, a gene involved in the establishment of asymmetry in early C. elegans embryos, mediates the asymmetric localization of PAR-3. Development 122, 3133–3140 (1996).
Goehring, N. W. et al. Polarization of PAR proteins by advective triggering of a pattern-forming system. Science 334, 1137–1141 (2011).
Cuenca, A. A., Schetter, A., Aceto, D., Kemphues, K. & Seydoux, G. Polarization of the C. elegans zygote proceeds via distinct establishment and maintenance phases. Development 130, 1255–1265 (2003).
Zipperlen, P., Fraser, A. G., Kamath, R. S., Martinez-Campos, M. & Ahringer, J. Roles for 147 embryonic lethal genes on C.elegans chromosome I identified by RNA interference and video microscopy. EMBO J. 20, 3984–3992 (2001).
Piano, F. et al. Gene clustering based on RNAi phenotypes of ovary-enriched genes in C. elegans. Curr. Biol. 12, 1959–1964 (2002).
Sonnichsen, B. et al. Full-genome RNAi profiling of early embryogenesis in Caenorhabditis elegans. Nature 434, 462–469 (2005).
Piekny, A. J. & Mains, P. E. Rho-binding kinase (LET-502) and myosin phosphatase (MEL-11) regulate cytokinesis in the early Caenorhabditis elegans embryo. J. Cell Sci. 115, 2271–2282 (2002).
Willis, J. H., Munro, E., Lyczak, R. & Bowerman, B. Conditional dominant mutations in the Caenorhabditis elegans gene act-2 identify cytoplasmic and muscle roles for a redundant actin isoform. Mol. Biol. Cell 17, 1051–1064 (2006).
Rivers, D. M., Moreno, S., Abraham, M. & Ahringer, J. PAR proteins direct asymmetry of the cell cycle regulators Polo-like kinase and Cdc25. J. Cell Biol. 180, 877–885 (2008).
Baas, A. F. et al. Activation of the tumour suppressor kinase LKB1 by the STE20-like pseudokinase STRAD. EMBO J. 22, 3062–3072 (2003).
Kim, J. S., Hung, W., Narbonne, P., Roy, R. & Zhen, M. C. elegansSTRADα and SAD cooperatively regulate neuronal polarity and synaptic organization. Development 137, 93–102 (2010).
Tenlen, J. R., Molk, J. N., London, N., Page, B. D. & Priess, J. R. MEX-5 asymmetry in one-cell C. elegans embryos requires PAR-4- and PAR-1-dependent phosphorylation. Development 135, 3665–3675 (2008).
Spilker, A. C., Rabilotta, A., Zbinden, C., Labbe, J. C. & Gotta, M. MAP kinase signalling antagonizes PAR-1 function during polarization of the early Caenorhabditis elegans embryo. Genetics 183, 965–977 (2009).
Yoo, A. S., Bais, C. & Greenwald, I. Crosstalk between the EGFR and LIN-12/Notch pathways in C. elegans vulval development. Science 303, 663–666 (2004).
Matsubara, Y. et al. The adaptor-like protein ROG-1 is required for activation of the Ras-MAP kinase pathway and meiotic cell cycle progression in Caenorhabditis elegans. Gen. Cells 12, 407–420 (2007).
Green, R. A. et al. A high-resolution C. elegans essential gene network based on phenotypic profiling of a complex tissue. Cell 145, 470–482 (2011).
Wright, A. J. & Hunter, C. P. Mutations in a β-tubulin disrupt spindle orientation and microtubule dynamics in the early Caenorhabditis elegans embryo. Mol. Biol. Cell 14, 4512–4525 (2003).
Krueger, L. E., Wu, J. C., Tsou, M. F. & Rose, L. S. LET-99 inhibits lateral posterior pulling forces during asymmetric spindle elongation in C. elegans embryos. J. Cell Biol. 189, 481–495 (2010).
Panbianco, C. et al. A casein kinase 1 and PAR proteins regulate asymmetry of a PIP(2) synthesis enzyme for asymmetric spindle positioning. Dev. Cell 15, 198–208 (2008).
Collart, M. A. & Panasenko, O. O. The CCR4–NOT complex. Gene 492, 42–53 (2012).
DeBella, L. R., Hayashi, A. & Rose, L. S. LET-711, the Caenorhabditis elegans NOT1 ortholog, is required for spindle positioning and regulation of microtubule length in embryos. Mol. Biol. Cell 17, 4911–4924 (2006).
Schade, M. A., Reynolds, N. K., Dollins, C. M. & Miller, K. G. Mutations that rescue the paralysis of Caenorhabditis elegans ric-8 (synembryn) mutants activate the G α(s) pathway and define a third major branch of the synaptic signalling network. Genetics 169, 631–649 (2005).
Liu, J., Maduzia, L. L., Shirayama, M. & Mello, C. C. NMY-2 maintains cellular asymmetry and cell boundaries, and promotes a SRC-dependent asymmetric cell division. Dev. Biol. 339, 366–373 (2010).
Mayer, M., Depken, M., Bois, J. S., Julicher, F. & Grill, S. W. Anisotropies in cortical tension reveal the physical basis of polarizing cortical flows. Nature 467, 617–621 (2010).
Rose, L. S., Lamb, M. L., Hird, S. N. & Kemphues, K. J. Pseudocleavage is dispensable for polarity and development in C. elegans embryos. Dev. Biol. 168, 479–489 (1995).
Gally, C. et al. Myosin II regulation during C. elegans embryonic elongation: LET-502/ROCK, MRCK-1 and PAK-1, three kinases with different roles. Development 136, 3109–3119 (2009).
Kamath, R. S. et al. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421, 231–237 (2003).
Rual, J. F. et al. Toward improving Caenorhabditis elegans phenome mapping with an ORFeome-based RNAi library. Gen. Res. 14, 2162–2168 (2004).
Shannon, P. et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Gen. Res. 13, 2498–2504 (2003).
Raffel, M. Particle Image Velocimetry: a Practical Guide 2nd edn (Springer, 2007).
Acknowledgements
This work was supported by a Wellcome Trust Senior Research Fellowship (054523, to J.A.), postdoctoral fellowships from the Human Frontier Science Program (to B.T.F. and J.R.), a Herchel Smith Post-doctoral fellowship (to J.R.) and an EMBO fellowship (to B.T.F.). We thank P. Mains (Department of Biochemistry and Molecular Biology, University of Calgary, Canada) for providing let-502(ts) and mel-11(ts) mutants, and K. Kemphues (Department of Molecular Biology and Genetics, Cornell University, USA) for NMY-2-expressing bacteria. We thank F. Antigny, D. Lefer, V. Karabacak, S. Kroschwald, A. Maffioletti and A. Sayadian for their contribution in visual scoring and S. Fürthauer for help with hydrodynamic length measurements. We thank the media team of the Gurdon Institute for preparing screen reagents. We also thank C. Dix and R. Durbin for comments on the manuscript. Some nematode strains used in this work were provided by the Caenorhabditis Genetics Center, which is funded by the NIH National Center for Research Resources (NCRR). The P4A1 monoclonal antibody developed by J. Priess was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by The University of Iowa, Department of Biology, Iowa City, IA 52242.
Author information
Authors and Affiliations
Contributions
B.T.F. and J.R. designed and carried out the screen, analysed the network, characterized polarity candidates and drafted the manuscript. C.L. participated in the primary screen experiments. S.N. and S.G. designed and performed the biophysical analysis of myosin regulators. E.Z. generated the GFP::NOP-1 transgenic strain. T.I. and M.S. isolated the nmy-2(ts) and pkc-3(ts) mutants. J.A. participated in the design and coordination of the study and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 401 kb)
Supplementary Table 1
Supplementary Information (XLS 331 kb)
Supplementary Table 2
Supplementary Information (XLS 993 kb)
Supplementary Table 3
Supplementary Information (XLS 236 kb)
Supplementary Table 4
Supplementary Information (XLS 42 kb)
Supplementary Table 5
Supplementary Information (XLS 27 kb)
Supplementary Table 6
Supplementary Information (XLS 35 kb)
Supplementary Table 7
Supplementary Information (XLS 35 kb)
Time-lapse movie of NMY-2::GFP expressed in a wild-type control embryo.
Cortical projection of NMY-2::GFP in a wild-type control embryo. Images were acquired every 5 s. (MOV 1596 kb)
Time-lapse movie of NMY-2::GFP expressed in a cdc-42(RNAi) embryo.
Cortical projection of NMY-2::GFP in a cdc-42(RNAi) embryo. Images were acquired every 5 s. (MOV 1863 kb)
Time-lapse movie of NMY-2::GFP expressed in a csnk-1(RNAi) embryo.
Cortical projection of NMY-2::GFP in a csnk-1(RNAi) embryo. Images were acquired every 5 s. (MOV 1665 kb)
Time-lapse movie of NMY-2::GFP expressed in a mlc-5(RNAi) embryo.
Cortical projection of NMY-2::GFP in a mlc-5(RNAi) embryo. Images were acquired every 5 s. (MOV 2316 kb)
Time-lapse movie of NMY-2::GFP expressed in a unc-45(RNAi) embryo.
Cortical projection of NMY-2::GFP in a unc-45(RNAi) embryo. Images were acquired every 5 s. (MOV 1433 kb)
Time-lapse movie of NMY-2::GFP expressed in a Y54H5A.2(RNAi) embryo.
Cortical projection of NMY-2::GFP in a Y54H5A.2(RNAi) embryo. Images were acquired every 5 s. (MOV 3730 kb)
Time-lapse movie of NMY-2::GFP expressed in a plst-1(RNAi) embryo.
Cortical projection of NMY-2::GFP in a plst-1(RNAi) embryo. Images were acquired every 5 s. (MOV 1718 kb)
Time-lapse movie of NMY-2::GFP expressed in a unc-59(RNAi) embryo.
Cortical projection of NMY-2::GFP in a unc-59(RNAi) embryo. Images were acquired every 5 s. (MOV 1427 kb)
Time-lapse movie of NMY-2::GFP expressed in a erm-1(RNAi) embryo.
Cortical projection of NMY-2::GFP in a erm-1(RNAi) embryo. Images were acquired every 5 s. (MOV 1252 kb)
Time-lapse movie of NMY-2::GFP expressed in a cnt-2(RNAi) embryo.
Cortical projection of NMY-2::GFP in a cnt-2(RNAi) embryo. Images were acquired every 5 s. (MOV 1864 kb)
Time-lapse movie of NMY-2::GFP expressed in a gsp-1(RNAi) embryo.
Cortical projection of NMY-2::GFP in a gsp-1(RNAi) embryo. Images were acquired every 5 s. (MOV 1642 kb)
Time-lapse movie of NMY-2::GFP expressed in a nop-1(RNAi) embryo.
Cortical projection of NMY-2::GFP in a nop-1(RNAi) embryo. Images were acquired every 5 s. (MOV 1495 kb)
Rights and permissions
About this article
Cite this article
Fievet, B., Rodriguez, J., Naganathan, S. et al. Systematic genetic interaction screens uncover cell polarity regulators and functional redundancy. Nat Cell Biol 15, 103–112 (2013). https://doi.org/10.1038/ncb2639
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2639
This article is cited by
-
Benchmarking selected computational gene network growing tools in context of virus-host interactions
Scientific Reports (2017)
-
The polarity protein Scrib mediates epidermal development and exerts a tumor suppressive function during skin carcinogenesis
Molecular Cancer (2015)
-
A sensitised RNAi screen reveals a ch-TOG genetic interaction network required for spindle assembly
Scientific Reports (2015)
-
ZIPping to Pain Relief: The Role (or Not) of PKMζ in Chronicc Pain
Molecular Pain (2013)